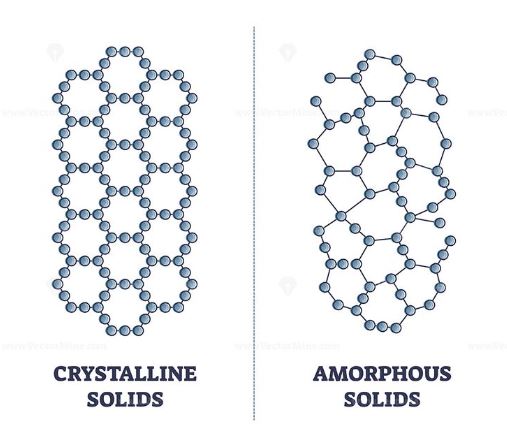
Solids are one of the fundamental states of matter, characterized by their definite shape and volume. They are typically classified into two main categories based on their atomic structure: crystalline and amorphous. Crystalline solids, like diamonds and table salt, have a highly organized atomic structure with a repeating pattern extending throughout the material. This regularity imparts specific properties to these materials, such as well-defined melting points and varying physical properties depending on the direction in which they are measured.
In contrast, amorphous solids, sometimes referred to as non-crystalline solids, lack this long-range order. This absence of a repeating atomic arrangement leads to different properties and behaviors. The question then arises: “What is an amorphous solid?” To answer this, we need to delve into their defining features and how they differ from their crystalline counterparts.
What is an Amorphous Solid?
An amorphous solid is a type of solid material that does not possess a well-defined, repeating atomic structure. Unlike crystalline solids, which have an orderly, periodic arrangement of atoms extending throughout the material, amorphous solids have a more disordered atomic arrangement. This means that while there may be some local order, there is no long-range periodicity in the structure.
The key distinction between amorphous and crystalline solids lies in this structural organization. In crystalline solids, the atoms are arranged in a regular lattice that extends indefinitely in all directions, giving rise to properties such as anisotropy, where physical properties vary with direction. In contrast, amorphous solids lack this long-range order, resulting in isotropy, where properties are consistent regardless of direction.
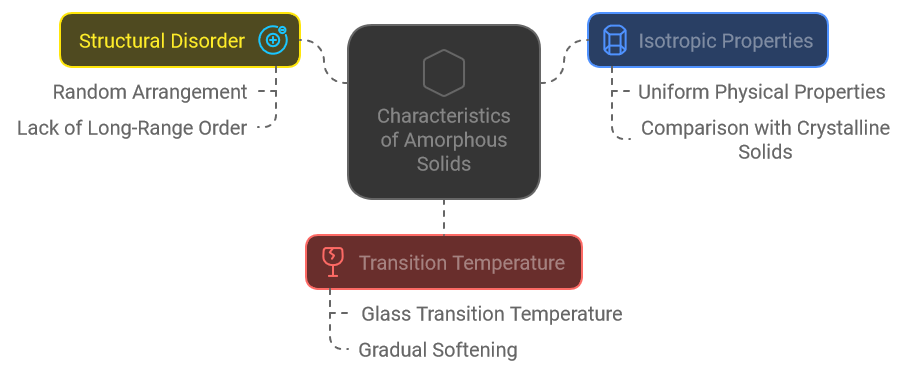
Characteristics of Amorphous Solids
Structural Disorder
Amorphous solids are characterized by their structural disorder. Unlike the orderly lattice found in crystalline materials, the atoms or molecules in amorphous solids are arranged in a random or irregular pattern. This disorganization means there are no repeating units that extend over long distances, leading to a more chaotic atomic arrangement.
Isotropic Properties
Due to their lack of long-range order, amorphous solids typically exhibit isotropic properties. This means that their physical properties, such as strength and thermal conductivity, are the same in all directions. This contrasts with crystalline solids, where properties can vary based on the direction of measurement due to the directional nature of their atomic arrangement.
Transition Temperature
Amorphous solids do not have a distinct melting point as crystalline solids do. Instead, they have a characteristic temperature known as the glass transition temperature (Tg). At this temperature, the material transitions from a hard and brittle state to a more rubbery or fluid state. This transition does not involve a phase change but rather a gradual softening, reflecting the different behavior of amorphous solids compared to their crystalline counterparts.

Examples of Amorphous Solids
Glass
One of the most recognizable examples of an amorphous solid is glass, particularly traditional soda-lime glass. Soda-lime glass is used in a variety of everyday items, such as windows, bottles, and containers. Unlike crystalline materials, soda-lime glass does not have a regular atomic structure. Instead, its atoms are arranged in a random, disordered fashion. This lack of long-range order is what classifies it as an amorphous solid. Despite its disordered atomic arrangement, glass has many useful properties, such as transparency and a smooth surface, which make it a valuable material in various applications.
Plastics
Many common synthetic polymers are also examples of amorphous solids. Polystyrene and polycarbonate are two widely used types of plastic. Polystyrene, found in products like disposable cutlery and CD cases, and polycarbonate, used in items such as eyewear lenses and electronic casings, both exhibit amorphous structures. These materials have disordered atomic arrangements, which contribute to their unique properties, such as flexibility and impact resistance. Their amorphous nature allows them to be molded into various shapes and used in diverse applications.
Gels
Gels are another example of amorphous solids. They are composed of a network-like structure of polymer chains that are dispersed in a liquid, giving them their characteristic semi-solid consistency. Gelatin, found in foods like jelly and gummy candies, is a classic example. While gels exhibit some network order on a local scale, they do not have the long-range atomic order seen in crystalline materials. This network structure gives gels their ability to retain shape and moisture while remaining flexible.
Thin Films
In technology, amorphous solids are commonly used in thin films. These films are often found in devices like solar panels and display screens. For instance, amorphous silicon is used in thin-film solar cells due to its ability to absorb light and convert it into electricity, even though it lacks a crystalline structure. Similarly, amorphous materials are used in various display technologies to create uniform layers that contribute to the performance and efficiency of the devices.
Scientific Explanation
Amorphous solids form when materials cool rapidly from a molten state, preventing the atoms from arranging themselves into a regular crystalline lattice. This rapid cooling process, often referred to as quenching, does not allow sufficient time for the atoms to organize into a structured pattern. Instead, they become “frozen” in a disordered state, resulting in an amorphous solid. The lack of time for crystal formation during cooling is crucial to the development of amorphous solids, leading to their unique properties compared to crystalline materials.
Conclusion
Amorphous solids are a fascinating class of materials distinguished by their lack of long-range atomic order. From everyday items like glass and plastics to advanced technologies such as thin-film solar panels, their unique properties make them highly valuable in various fields. Understanding the characteristics and formation of amorphous solids is important not only for scientific inquiry but also for practical applications that impact many aspects of modern life. Whether it’s the transparency of glass or the flexibility of plastics, the amorphous nature of these materials plays a crucial role in their functionality and utility.
FAQs
1. What is the main difference between amorphous and crystalline solids?
Amorphous solids lack a long-range, repeating atomic structure, whereas crystalline solids have a highly organized and repeating atomic lattice. This structural difference leads to variations in properties such as melting points and mechanical behavior. Crystalline solids exhibit anisotropic properties, meaning their physical properties vary with direction, while amorphous solids are isotropic, having consistent properties in all directions.
2. Why do amorphous solids not have a distinct melting point?
Amorphous solids do not have a distinct melting point because their atomic structure lacks the long-range order found in crystalline solids. Instead of a sharp melting point, amorphous solids have a glass transition temperature (Tg), which is the temperature at which the material transitions from a hard and brittle state to a more rubbery or fluid state.
3. Can you provide examples of common amorphous solids?
Yes, common examples of amorphous solids include:
- Glass: Used in windows, bottles, and various containers.
- Plastics: Such as polystyrene (found in disposable cutlery) and polycarbonate (used in eyewear lenses).
- Gels: Like gelatin in foods and cosmetic products.
- Thin Films: Found in technologies like solar panels and display screens.
4. How do amorphous solids form?
Amorphous solids typically form when a material cools rapidly from a molten state. This rapid cooling prevents the atoms from arranging into a regular crystalline structure, resulting in a disordered atomic arrangement. This process is known as quenching.
5. What are isotropic properties and why are they important in amorphous solids?
Isotropic properties refer to the characteristic of a material where its physical properties are the same in all directions. In amorphous solids, the lack of long-range atomic order results in isotropic properties, making them consistent regardless of the direction in which they are measured. This is in contrast to crystalline solids, which exhibit anisotropic properties with direction-dependent behavior.
6. Why are thin films made from amorphous materials used in technology?
Thin films made from amorphous materials, such as amorphous silicon, are used in technology because they can be deposited in very thin layers and still perform effectively. For example, amorphous silicon is used in thin-film solar cells because it can absorb light and convert it into electricity efficiently, despite lacking a crystalline structure. Similarly, in display technologies, amorphous materials help create uniform layers that enhance performance.
7. What is the significance of the glass transition temperature (Tg) in amorphous solids?
The glass transition temperature (Tg) is significant because it marks the temperature at which an amorphous solid transitions from a rigid and brittle state to a more flexible, rubbery, or fluid state. Unlike crystalline solids, which have a sharp melting point, the Tg reflects the gradual softening of the material and is crucial for understanding its thermal properties and behavior in various applications.